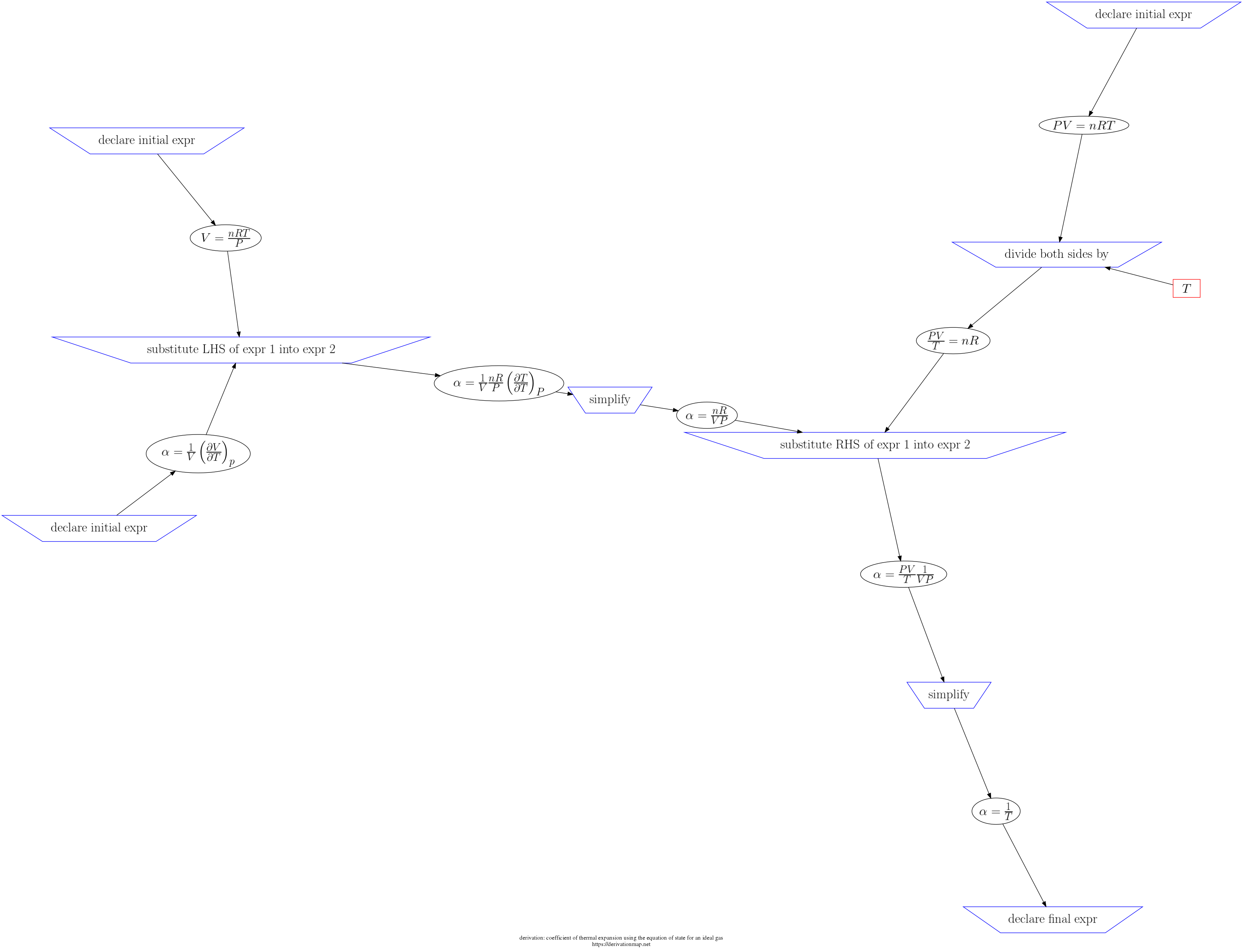
This page contains three views of the steps in the derivation: d3js, graphviz PNG, and a table.
Notes for this derivation:
https://notendur.hi.is/hj/EE2/HD1lausn.pdf
| Index | Inference Rule | Input latex | Feeds latex | Output latex | step validity | dimension check | unit check | notes |
|---|---|---|---|---|---|---|---|---|
| 8 | simplify |
|
|
|
LHS diff is 0 RHS diff is (pdg7586*pdg8134 - 1)/pdg7343 |
6925244346:
2472653783: |
6925244346:
2472653783: |
|
| 5 | declare initial expr |
|
|
|
no validation is available for declarations |
8435841627:
|
8435841627:
|
|
| 2 | declare initial expr |
|
|
|
no validation is available for declarations |
3497828859:
inconsistent dimensions |
3497828859:
N/A |
|
| 7 | substitute RHS of expr 1 into expr 2 |
|
|
|
LHS diff is 0 RHS diff is (-pdg7586*pdg8134 + 1)/pdg7343 |
2613006036:
5962145508: 6925244346: |
2613006036:
5962145508: 6925244346: |
|
| 1 | declare initial expr |
|
|
|
no validation is available for declarations |
3464107376:
error for dim with 3464107376 |
3464107376:
N/A |
|
| 4 | simplify |
|
|
|
valid |
1311403394:
error for dim with 1311403394 5962145508: |
1311403394:
N/A 5962145508: |
|
| 6 | divide both sides by |
|
|
|
valid |
8435841627:
2613006036: |
8435841627:
2613006036: |
|
| 9 | declare final expr |
|
|
|
no validation is available for declarations |
2472653783:
|
2472653783:
|
|
| 3 | substitute LHS of expr 1 into expr 2 |
|
|
|
LHS diff is 0 RHS diff is -pdg2834*pdg8179/(pdg7586*pdg8134) + 1/pdg7343 |
3497828859:
inconsistent dimensions 3464107376: error for dim with 3464107376 1311403394: error for dim with 1311403394 |
3497828859:
N/A 3464107376: N/A 1311403394: N/A |
| symbol ID | category | latex | scope | dimension | name | value | Used in derivations | references | |
|---|---|---|---|---|---|---|---|---|---|
| 8134 | variable |
P
\(P\) |
real |
|
pressure | 13 | |||
| 8179 | constant |
R
\(R\) |
real |
|
ideal gas constant |
8.31446261815324 J⋅K^{−1} mol^{−1} |
8 | ||
| 2834 | variable |
n
\(n\) |
real | dimensionless | amount of substance | 8 | |||
| 4686 | variable |
\alpha
\(\alpha\) |
real |
|
expansion coefficient | 6 | |||
| 7586 | variable |
V
\(V\) |
real |
|
volume | 15 | |||
| 7343 | variable |
T
\(T\) |
real |
|
temperature | 18 |